Spotlight
Letter from Dr. Stephen I. Katz: Update on the NIAMS Intramural Research Program
Dear Colleagues:
Approximately 10 percent of the National Institutes of Health (NIH), and in turn NIAMS’, annual budget is devoted to research conducted right here in Bethesda, Maryland, through our Intramural Research Program (IRP). This investment is yielding high returns, and I would like to take this opportunity to tell you more about our IRP and the talented group of scientists who work in it.
The intramural research program at NIH, comprised of scientists from 23 Institutes and Centers, provides a stimulating environment where long-term, high-impact projects can be pursued. The diversity of scientists and research interests across the NIH provides a unique incubator where truly innovative solutions can be explored. Here at the NIAMS, our IRP is home to 21 principal investigators, led by Scientific Director John O’Shea, M.D., and Clinical Director Richard Siegel, M.D., Ph.D. Our program spans eight primary focus areas, including bone biology, clinical and epidemiological research, genomics and epigenomics, immunology, muscle biology, skin biology, structural biology, and stem cell and induced pluripotent stem cell biology. Our intramural researchers’ work is frequently published in top-tier journals, including The New England Journal of Medicine, Science, Nature and Cell, and they train numerous postdoctoral research fellows and students, helping to build the next generation of researchers.
Image: Stephen I. Katz, M.D., Ph.D.
News
 NIH, Industry and Nonprofits Join Forces to Speed Validation of Disease Targets: Goal Is To Develop New Treatments Earlier, Beginning With Alzheimer’s, Type 2 Diabetes, and Autoimmune Disorders
NIH, Industry and Nonprofits Join Forces to Speed Validation of Disease Targets: Goal Is To Develop New Treatments Earlier, Beginning With Alzheimer’s, Type 2 Diabetes, and Autoimmune Disorders
The NIH, 10 biopharmaceutical companies and several nonprofit organizations launched an unprecedented partnership to transform the current model for identifying and validating the most promising biological targets of disease for new diagnostics and drug development.
Protein Replacement Therapy Shows Promise in Treating Rare Skin Disorder
Replacing a protein that is crucial to ensuring that the skin’s epidermis and dermis adhere to one another may be the key to treating a debilitating skin disorder effectively, according to research conducted in mice and funded in part by the NIAMS.
Fingernails Yield Clues to Limb Regeneration
New research funded in part by the NIAMS has uncovered chemical signals that drive the regeneration of lost digit tips in mice. The findings, published in the journal Nature, could eventually lead to the development of novel treatments for amputees.
Image: Regenerated mouse toe tip (left); inactivated mouse toe signaling pathway
Photo Credit: Mayumi Ito
New Reforms to Federal Grant Policies
Last year, the Office of Management and Budget (OMB) solicited grantee and stakeholder comments, and in December, released new grant policy guidance, “Uniform Administrative Requirements, Cost Principles, and Audit Requirements for Federal Awards.”
NIH Director’s Blog
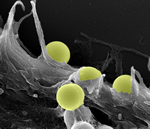
Snapshots of Life: Nanotechnology Meets Cell Biology
Many of the most exciting frontiers in biomedical research sound like the stuff of science fiction, but here is some work that even looks like it’s straight from the set of “Star Trek”! This scanning electron micrograph captures the pivotal moment when nanospheres—a futuristic approach to drug delivery—are swallowed up by a human fibroblast cell.
Image: Photo credit: Matthew Ware and Biana Godin Vilentchouk, Houston Methodist Research Institute, Texas
Other Federal News
Agency for Healthcare Research and Quality (AHRQ) Key Questions Available for Comment until March 3: Pain Management Injection Therapies for Low-back Pain
AHRQ's Technology Assessment Program uses state-of-the-art methodologies for assessing the clinical utility of medical interventions. Technology assessments are based on a systematic review of the literature, along with appropriate qualitative and quantitative methods of synthesizing data from multiple studies.
FDA Launches Advisory Committee Membership Nomination Portal
The U.S. Food and Drug Administration (FDA) today launched the Advisory Committee Membership Nomination Portal, an online, interactive system that allows interested individuals to submit nominations for membership to any of the agency’s 33 advisory committees.
Some Wart Removers Are Flammable
Some cryogenic wart removers, which remove warts from the skin by freezing them off, have caught fire during use at home, harming consumers or setting fire to items around the house. Since 2009, the FDA, which regulates wart removers as medical devices, has received 14 such reports about over-the-counter wart remover products, which are a mixture of liquid dimethyl ether and propane.
NEW PUBLICATIONS
NIH Research Matters
NIH Research Matters is a review of NIH research from the Office of Communications and Public Liaison, Office of the Director, NIH.
Arthritis Genetics Analysis Aids Drug Discovery
An international research team identified 42 new areas in the human genome associated with rheumatoid arthritis—and found that many are already the targets of drugs approved for other conditions. The findings hint at new treatment approaches for the disease.
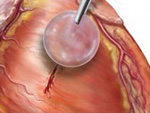
Nature-Inspired Surgical Glue Mends Hearts
Researchers developed a new tissue adhesive that is biodegradable, biocompatible and easily manipulated. It could allow for less invasive surgeries that don’t require sutures or staples.
Image: Glue secures patches to seal holes in beating hearts. Image courtesy of Karp Laboratory
NIH News in Health
Read practical health information in NIH News in Health, which is reviewed by the NIH’s medical experts and is based on research conducted either by the NIH’s own scientists or by its grantees at universities and medical schools around the country.
Gripped by Gout: Avoiding the Ache and Agony
Sudden, painful swelling at the base of the big toe is often the first warning sign of gout. It can affect other joints as well. Without treatment, gout can lead to severe joint damage and make it hard for you to move. The good news is most types of gout are treatable, especially if caught early.
MEETINGS
FDA Public Meeting on Fibromyalgia Patient-Focused Drug Development
This is a rescheduling of the original December 10, 2013 meeting.
March 26, 2014
1–5 p.m.
FDA White Oak Campus
The Great Room (Room 1503A)
10903 New Hampshire Avenue
Silver Spring, Maryland 20903
NIH Wednesday Afternoon Lecture Series
The NIH’s Wednesday Afternoon Lecture Series offers weekly lectures every Wednesday at 3 p.m. in Masur Auditorium, Building 10, NIH Campus. Renowned scientists from around the globe present research on a variety of topics. The lectures are Continuing Medical Education-certified, open to the public and available live via webcast.
NIH Science Lectures and Events Available Online
The NIH hosts a number of science seminars and events that are available online through real-time streaming video. You can watch an event at your convenience as an on-demand video or a downloadable podcast. Most events are available to all; a few are broadcast for the NIH or the U.S. Department of Health and Human Services and are marked as such. See additional details on events.
NIAMS Exhibit Schedule
The NIAMS exhibit is traveling to several events. See the schedule of health fairs and exhibits.
Image: The NIAMS Exhibit
FUNDING ANNOUNCEMENTS
NIAMS Announcements
Career Development Award in Biomedical Big Data Science for Clinicians and Doctorally Prepared Scientists (K01)
(RFA-HG-14-007)
Letter of Intent Receipt Dates: 30 days before the application due date
Application Receipt Dates: April 1, 2014; April 1, 2015
Courses for Skills Development in Biomedical Big Data Science (R25)
(RFA-HG-14-008)
Letter of Intent Receipt Dates: 30 days before the application due date
Application Receipt Dates: April 1, 2014; April 1, 2015; and April 1, 2016
Open Educational Resources for Biomedical Big Data (R25)
(RFA-HG-14-009)
Letter of Intent Receipt Dates: 30 days before the application due date
Application Receipt Dates: April 1, 2014; April 1, 2015; and April 1, 2016
PHS 2014-02 Omnibus Solicitation of the NIH, CDC, FDA and ACF for Small Business Innovation Research Grant Applications (Parent SBIR [R43/R44])
(PA-14-071)
Letter of Intent Receipt Date: Not applicable
Application Receipt Dates: Standard dates apply
PHS 2014-02 Omnibus Solicitation of the NIH for Small Business Technology Transfer Grant Applications (Parent STTR [R41/R42]
(PA-14-072)
Letter of Intent Receipt Date: Not applicable
Application Receipt Dates: Standard dates apply
Administrative Supplements to Existing NIH Grants and Cooperative Agreements (Parent Admin Supp)
(PA-14-077)
Letter of Intent Receipt Date: Not applicable
Application Receipt Date: No due date. However, NIAMS is not accepting applications from July 1, 2014, to October 1, 2014.
Change of Grantee Organization (Type 7 Parent)
(PA-14-078)
Letter of Intent Receipt Date: Not applicable
Application Receipt Date: A change of grantee organization request must be made before the anticipated start date at the new organization and preferably several months in advance.
Successor-in-Interest (Type 6 Parent)
(PA-14-079)
Letter of Intent Receipt Date: Not applicable
Application Receipt Date: A change of grantee organization request must be made before the anticipated start date at the new organization and preferably several months in advance.
NIH Common Fund Initiative Announcements
NIH Directors Biomedical Research Workforce Innovation Award: Broadening Experiences in Scientific Training (BEST) (DP7)
(RFA-RM-13-019)
Letter of Intent Receipt Date: February 28, 2014
Application Receipt Date: March 31, 2014
Other Funding Announcements
Limited Competition: Mutant Mouse Resource and Research Centers (U42)
(RFA-OD-14-003)
Letter of Intent Receipt Date: Not applicable
Application Receipt Date: May 1, 2014
Notice of Intent to Publish a Funding Opportunity Announcement for Mobilizing Research: A Research Resource to Enhance mHealth Research (U2C)
(NOT-OD-14-035)
NIH Operates Under a Continuing Resolution
(NOT-OD-14-043)